|
Towards the objectives of the UnitedStates Food and Drug Administration (FDA) generic drug science and research program, it is of vital importance in developing product-specific guidances (PSGs) with recommendations that can facilitate and guide generic product development. 3School of Biomedical Engineering, Science and Health Systems, Drexel University, Philadelphia, PA, United States.2Office of Research and Standards, Office of Generic Drugs, Center for Drug Eval uation and Research, United States Food and Drug Administration, Silver Spring, MD, United States.1College of Computing and Informatics, Drexel University, Philadelphia, PA, United States. 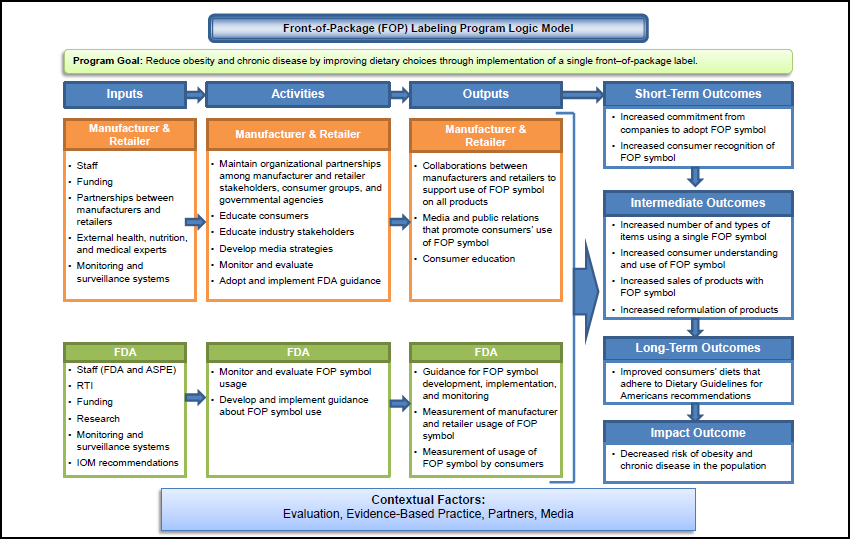 
Unapproved Drugs: Drugs Marketed in the United States that Do Not Have Required FDA Approval, where information about unapproved animal drugs products is available.Yiwen Shi 1, Ping Ren 2, Yi Zhang 2, Xiajing Gong 2, Meng Hu 2 and Hualou Liang 3*. 
Unapproved Drugs: Drugs Marketed in the United States that Do Not Have Required FDA Approval, where information about unapproved human drugs products is available.Office of Nonprescription Products, where information about human OTC drug products is available.Biologics Products and Establishments, where information about vaccines, allergenics, and blood products is available.Drugs FDA, where information about FDA-approved human brand name and generic drugs as well as therapeutic biological products is available.Animal and Veterinary Products, where information about animal drugs products is available.Information about animal and human drug products can be found on these FDA Web pages: Health information suppliers and others can download all of the electronic files companies have submitted from the National Library of Medicine’s Download Labels site. You can search for labels by drug name and link to the Library’s information resources about marketed drugs. The labels are also available on the National Library of Medicine's DailyMed web site. Proprietary Name SearchĪpplication Number or Regulatory Citation Search The device labeling on this website may not be the labeling on currently distributed products. The device labeling has been reformatted to make it easier to read but its content has not been altered nor verified by FDA. FDA has not reviewed this information prior to posting on this website. The device labeling and other device-specific information on this website have been voluntarily submitted to the FDA by device manufacturers. 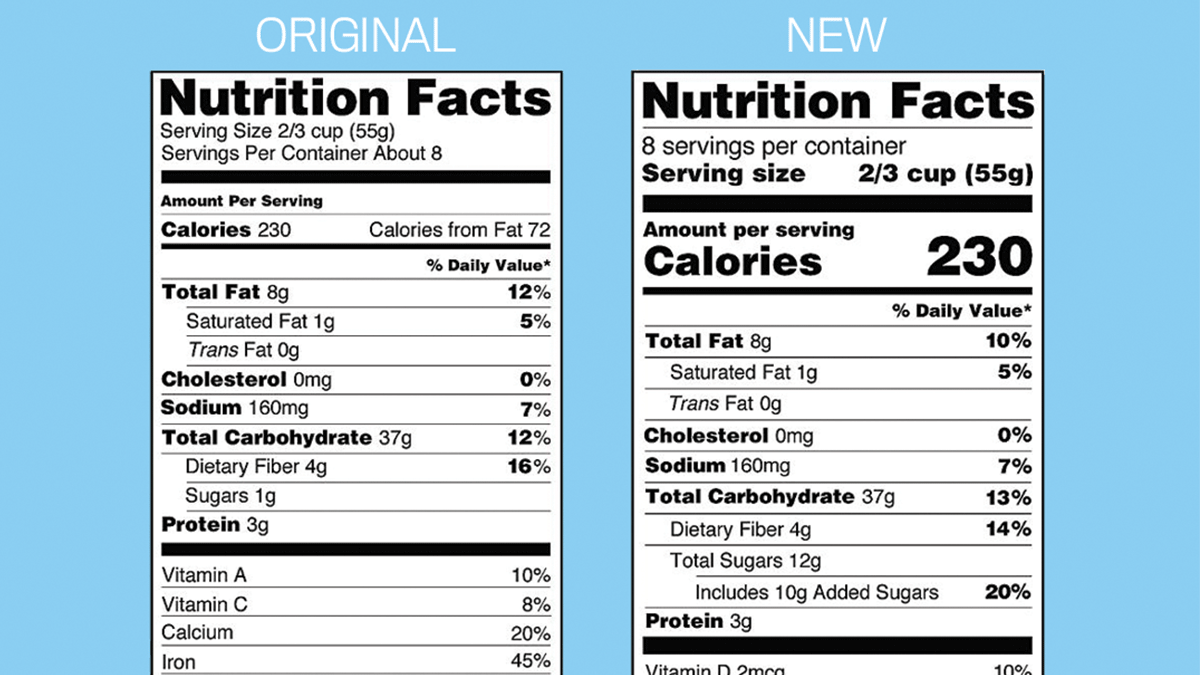
In addition, FDA is not aware of scientific evidence to support homeopathy as effective. Drugs marked "unapproved medical gas", "unapproved homeopathic" or "unapproved drug other" on this Web site have not been evaluated by FDA for safety and efficacy and their labeling has not been approved. Drugs marked "OTC monograph final" or "OTC monograph not final" are not checked for conformance to the monograph. Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies described in monographs. The drug labeling on this Web site may not be the labeling on currently distributed products or identical to the labeling that is approved. (See 21 CFR part 207.) The drug labeling and other information has been reformatted to make it easier to read but its content has neither been altered nor verified by FDA. The drug labels and other drug-specific information on this Web site represent the most recent drug listing information companies have submitted to the Food and Drug Administration (FDA). Please be aware of the following when using information from this Web site:
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
